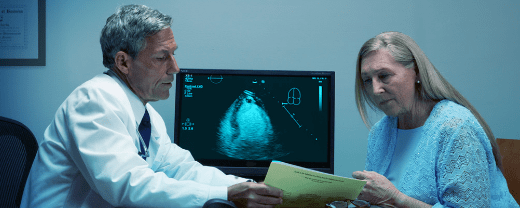
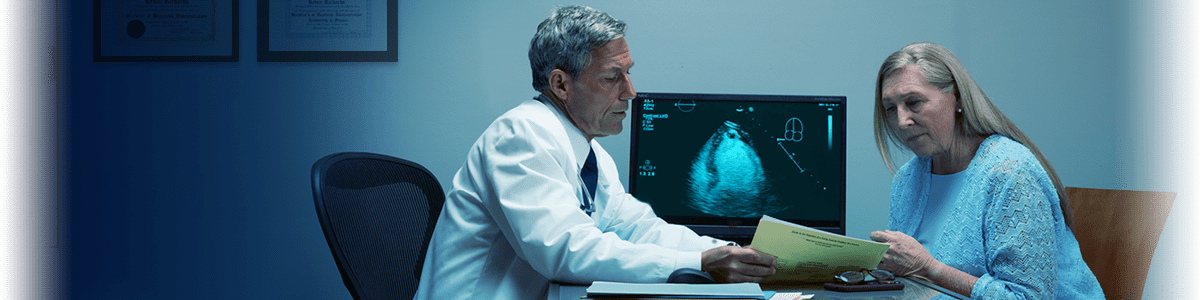
The most utilized ultrasound enhancing agent is approved for use in pediatric patients
Data on file, Lantheus.
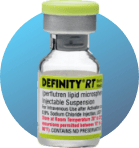
Store at Room Temperature (68°‑77°F; 20°‑25°C)

Activate in VIALMIX® RFID
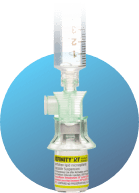
Attach ViaLok® Add 1.4 mL preservative-free Sodium Chloride Injection, USP
Swirl Rapidly 10 seconds


If the product is not used within 5 minutes of dilution, resuspend the microspheres by rapidly swirling the upright vial for 10 seconds before the product is withdrawn in the syringe.
Activated DEFINITY® RT may be used for up to 4 hours from the time of dilution, stored at room temperature in the product vial with the ViaLok® attached. A sterile syringe or cap should be attached to the ViaLok® until use. When ready to use, resuspend the microspheres by rapidly swirling the upright vial for 10 seconds before the product is withdrawn in the syringe.
DEFINITY® RT offers multiple dosing and administration options to meet patient- and practice‑specific needs.1
DILUTED BOLUS
CONTINUOUS INFUSION
BOLUS
DEFINITY® RT mixed with preservative-free 0.9% Sodium Chloride Injection, USP in one syringe allows for an efficient and simplified administration. This saves time, avoids the need for a 0.9% Sodium Chloride Injection, USP flush, and eliminates use of additional supplies and waste.
For more details on the diluted bolus method, watch this instructional video.
A continuous flow of DEFINITY® RT combined with preservative-free 0.9% Sodium Chloride Injection, USP provides a consistent, steady enhancement. This method is well-suited for studies performed over an extended period.
For more details on the continuous infusion, watch this instructional video.
DEFINITY® RT may be administered as a simple, straight bolus injection followed by a 0.9% Sodium Chloride Injection, USP flush to offer an easy, rapid image enhancement in small doses.
Smaller, incremental dose amounts of 0.2 mL to 0.3 mL are better suited for current ultrasound system technology. The maximum allowable dose is 20 μL/kg.
For more details on the bolus method, watch this instructional video.
References:
DEFINITY® RT [package insert]. N. Billerica, MA: Lantheus Medical Imaging, Inc.
VialMix® RFID User's Guide. N. Billerica, MA: Lantheus Medical Imaging, Inc.
Sboros V, Moran CM, Pye SD, McDicken WN. Contrast Agent Stability: A Continuous B-Mode Imaging Approach. Ultrasound in Med & Biol. 2001;27(10):1367-1377.